RESEARCH
Corepressors and DNA damage
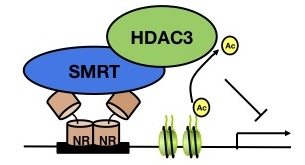
Transcriptional regulation is a critical process that can be both positively and negatively regulated, and this regulation is influenced by coactivators and corepressors. The corepressor silencing mediator for retinoid and thyroid receptors (SMRT) interacts with various nuclear receptors and transcription factors and mediates repression by recruiting histone deacetylase 3 (HDAC3) among other components.
Work on these factors revealed a novel interaction between SMRT and DNA-dependent protein kinase complex (DNA-PK), a DNA repair complex. This complex is composed of the kinase DNA-PKcs and the Ku70 and Ku80 subunits. The DNA-PK complex plays a critical role in DNA repair, and one of its subunits, Ku70, is important for regulating apoptosis. In my master's work in the Lazar lab at the University of Pennsylvania, my collaborators and I characterized the interaction of these proteins and assessed the functionality of this interaction. (Yu, et. al, 2006)
|
Figure 1. Model The corepressor SMRT interacts with the histone deacetylase HDAC3, leading to deacetylation and compaction of DNA. |
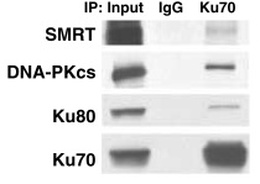
Figure 2. SMRT interacts with the DNA-PK repair complex
SMRT coimmunoprecipitates with components of the DNA-PK repair complex. Cell lysate from 293T cells was incubated with antibody to the Ku70 subunit and western blots were probed with the indicated antibodies. |